Заглавная страница Избранные статьи Случайная статья Познавательные статьи Новые добавления Обратная связь КАТЕГОРИИ: ТОП 10 на сайте Приготовление дезинфицирующих растворов различной концентрацииТехника нижней прямой подачи мяча. Франко-прусская война (причины и последствия) Организация работы процедурного кабинета Смысловое и механическое запоминание, их место и роль в усвоении знаний Коммуникативные барьеры и пути их преодоления Обработка изделий медицинского назначения многократного применения Образцы текста публицистического стиля Четыре типа изменения баланса Задачи с ответами для Всероссийской олимпиады по праву 
Мы поможем в написании ваших работ! ЗНАЕТЕ ЛИ ВЫ?
Влияние общества на человека
Приготовление дезинфицирующих растворов различной концентрации Практические работы по географии для 6 класса Организация работы процедурного кабинета Изменения в неживой природе осенью Уборка процедурного кабинета Сольфеджио. Все правила по сольфеджио Балочные системы. Определение реакций опор и моментов защемления |
Boiling Point and Melting Point
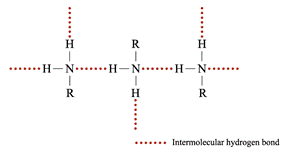
1° or 2° amines can form strong hydrogen bonds with each other Þ the b.p. is higher than those of alkanes but lower than those of alcohols of comparable molecular masses ∵ O – H bond is more polar than N – H bond as oxygen is more electronegative Þ stronger hydrogen bonds are formed between alcohol molecules
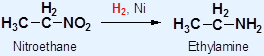
3° amines have lower b.p. and m.p. than 1° and 2° all hydrogen in 3° amines are replaced by organic groups Þ no hydrogen bond can be formed Density Molecular mass of amines Þ densities Solubility • The solubility of amines in water: ∵ The intermolecular hydrogen bonds formed between 2° or 3° amines and water molecules decrease • Amines are soluble in common organic solvents, e.g. hexane, ethanol and propanone Preparation of Amines Hydrogen reduces a nitro compounds to a 1° amine in the presence of nickel catalyst
Example:
Reduction of Nitriles Reaction with Hydrogen Hydrogen reduces a nitrile to a 1° amine in the presence of nickel catalyst
Example:
Reaction with Lithium Tetrahydridoaluminate Nitriles are reduced to 1° amines by LiAlH4 in dry ether
Example:
Reduction of Amides Reaction with Lithium Tetrahydridoaluminate Amides can be reduced by LiAlH4 to give 1°, 2° or 3° amines
• The product is 1° amine if R’ and R’’ = H • The product is 2° amine if R’ or R’’ = H Examples:
Hofmann Degradation • Amides with no substituent on the nitrogen react with Br2 in NaOH (or Cl2 in NaOH) to give amines • The amines formed have one carbon less than the parent amides
Reaction of Ammonia with Haloalkanes 1°, 2°, 3° and quaternary ammonium salts can be prepared by heating haloalkanes with ammonia under pressure
Examples:
• This method always give a mixture of products • Successive alkylations always occur because aliphatic amines are more reactive than ammonia Preparation of Phenylamine Reduction of Nitrobenzene (Zinin’s reactin) The reduction of the nitro group can be carried out by • Catalytic hydrogenation
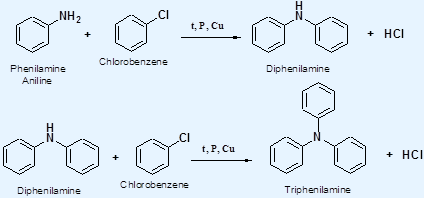
Reaction of Ammonia with Halobenzenes 1° phenylammines can be prepared by heating halobenzenes with ammonia under pressure
2° and 3° phenylammines can be prepared by heating halobenzenes with phenylamine under pressure
Reactions of Amines Basicity of Amines • Amines are far weaker bases than hydroxide ions but are stronger bases than water • The lone pair electrons on nitrogen atom can accept a proton from a water molecule Þ amines are basic
Amines dissolve in mineral acids such as HCl to form salts e.g.
Methylamine will partially dissociate into ions
pKb is used for convenience as Kb is small for weak bases
The basicity of amines is governed by the availability of the lone pair electrons on the nitrogen atom • Primary aliphatic amines are the most basic electron-releasing alkyl groups enhance the availability of the lone pair electrons on the nitrogen atom while ammonia does not • Aromatic amines are weaker bases than ammonia the lone pair electrons on the nitrogen atom are delocalized over the benzene ring making it less available to a proton
Acylation of amines
• 1° and 2° amines react readily in cold with ethanoyl chloride or anhydride to form N -substituted amides Examples:
• 1° and 2° amines react with benzoyl chloride in the presence of alkalis to form N -substituted amides Examples:
|
|||||||||||||||||
|
Последнее изменение этой страницы: 2017-02-07; просмотров: 307; Нарушение авторского права страницы; Мы поможем в написании вашей работы! infopedia.su Все материалы представленные на сайте исключительно с целью ознакомления читателями и не преследуют коммерческих целей или нарушение авторских прав. Обратная связь - 3.17.6.75 (0.005 с.) |














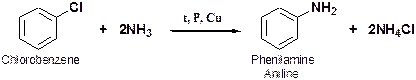




 The basic strength of amines is shown by the value of base dissociation constant (Kb)
The basic strength of amines is shown by the value of base dissociation constant (Kb)